Apart from superficial venous insufficiency, the major factor causing the occurrence and the development of clinical symptoms in chronic venous insufficiency is valve reflux in the deep venous system
[11] . Effective treatment of valve reflux requires replacement of the diseased valves. Autologous vein containing valves transplantation is the main treatment for deep venous valve replacement. Although transplantation is successful in correcting reflux and is associated with ulcer healing and the relief of symptoms, vein containing valves transplant can cause significant trauma to the patient and long-term valve competency rates and symptomatic control are poor
[12] . Problems arise prior to surgery since it is difficult to find a suitable donor valve. This is evidenced by the fact that 30%–40%
[13-14] of axillary vein valves, which are often used for superficial femoral venous valve replacement, are found to be incompetent prior to harvesting.
To overcome these limitations, artificial creation of implantable complex structures like vein valves has been pursued, and reported with encouraging results. However, the replacement has not been successfully tested in the long term and in addition; these are thrombogenic and require life-long anti-coagulation therapy
[11, 15-16] . Alternatively, tissue engineered vein containing valves can overcome these disadvantages. It has been reported that using tissue engineering, a tissue engineered vein containing valves constructed with self-derived endothelial cells and allogeneic acellular matrices, can provide the complex physiological valve structure and mechanical stability, but this elicited an immunogenic response
[5] .
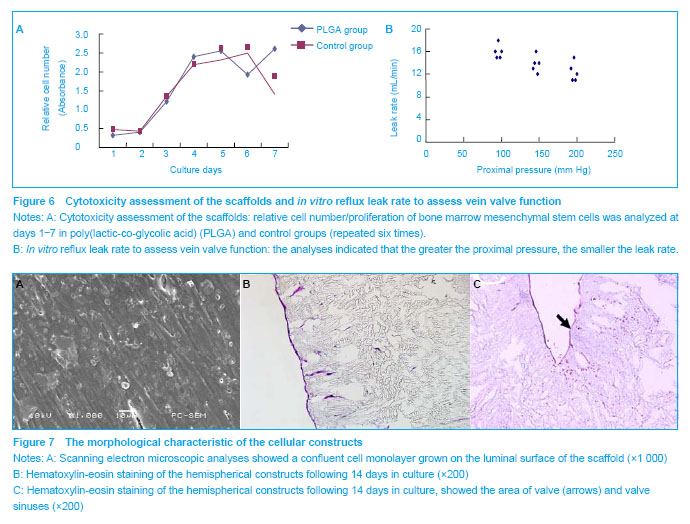
Due to the above limitations, a search for an ideal vein containing valves replacement continues. The major goal of our research group is to create a viable and functional vein containing valves, which has the advantages of the natural living tissue, including its resistance to thrombus formation and infection, as well as the ability to grow and remodel. In the present experiment, we have successfully produced a three-dimensional, porous scaffold from a thermoplastic PLGA that could then be used for tissue engineered venous valve structures. Scanning electron microscopic images and hematoxylin-eosin straining revealed that bone marrow mesenchymal stem cells which attached to the PLGA scaffold formed a near confluent layer. Tests showed that valve leaflets were movable when rinsed with PBS.Among a variety of issues, a major focus in tissue engineering is to design a suitable three-dimensional scaffold structure that can be a template for supporting cell growth, migration, differentiation and secretion of extracellular matrix proteins, as well as for directing the formation of new tissues during the tissue regeneration process
[17-18] . Ideally, scaffolds will be slowly resorbed in culture or following implantation, leaving only the tissue generated by the cells
[19] . Porosity and pore size of the scaffolds are also key factors
[20] , since a scaffold that has high porosity and large interconnected pores can enable mass transport, infiltration of cells, and interstitial flow fluid
[21-22] . As a popular biodegradable medical material, PLGA has been extensively investigated due to its good biocompatibility, controllable biodegradability, and good process ability
[23-26] . In this study, we successfully fabricated a porous PLGA scaffold. Tests have shown that PLGA could induce cell adhesion and proliferation, indicating the biocompatibility of the polymer. In addition, it is important that its degradation rate can be easily controlled by varying the copolymer ratio of lactic to glycolic acid
[27] . Since the valve and vein walls play different roles in venous return, it is expected that PLGA degradation and tissue invasion in valve and vein walls can proceed differently, which will be further studied in the future.
Although endothelial and smooth muscle cells have been considered to be the main seeding cells for tissue engineered vascular graft, they are harvested from intact vascular structures of the donor. Moreover, harvesting methods are traumatic; the harvested cells are limited in quantity and proliferation capacity. The limited proliferation potential of harvested cells makes it impossible to obtain large amounts of cells from a small vessel biopsy. Due to the fact that bone marrow mesenchymal stem cells have great potential in differentiating towards multiple cell lineages, they are widely utilized as an alternative cell source for tissue engineering. Bone marrow mesenchymal stem cells have been reported to be capable of differentiating into endothelial and vascular smooth muscle cells
[9-10, 28-29] . Bone marrow mesenchymal stem cells and endothelial cells performed similarly against platelet adhesion
[30] . These results strongly suggest that bone marrow mesenchymal stem cells can be used as an alternative cell source to endothelial cells for the prevention of thrombogenesis in vascular reconstruction. Some studies have found that bone marrow mesenchymal stem cells in the grafts could differentiate into vascular smooth muscle cells and endothelial cells in vivo
[31-32] . In the present study, we have shown that bone marrow mesenchymal stem cells have a great capacity for differentiating into multiple cell lineages, and PLGA can promote cell proliferation and differentiation. We have directly seeded bone marrow mesenchymal stem cells onto the scaffold, in order to differentiate into cells similar to native vessels in vitro and in vivo, and in vitro test have showed durability of cell adhesion is good.
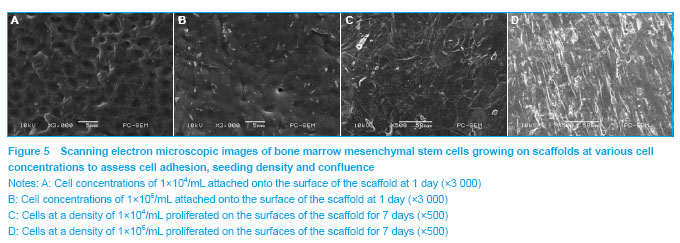
Cell seeding is the first step in tissue engineering approaches that incorporate cells into or onto scaffolds for culture or implantation. Since PLGA is hydrophobic, the porous scaffolds fabricated with these polymers float on the cell culture media and the scaffold does not absorb the culture media. Some approaches have been performed to improve the hydrophilicity of biodegradable polymer scaffolds and thus to ensure uniform and dense cell seeding. In our study, other than treating the scaffolds by pre-wetting with ethanol, we have customized a vacuum device. Prior to cell seeding, the scaffolds were placed in a vacuum flask, to allow its pores to be filled with culture media, and to increase seeding efficiency and reduce cell seeding time.
In summary, tissue engineered veins containing valves have been successfully constructed using a three-dimensional PLGA scaffold and bone marrow mesenchymal stem cells in vitro. Results of this study suggest that tissue engineered veins containing valves are potentially useful for the substitution and regeneration of vein valves, although further studies are warranted. Issues such as scaffold characteristics, patency, thrombosis, biodegradation speed and valve function should be assessed in long-term animal studies.

.jpg)
.jpg)